Rare Neurodegenerative Diseases
Neurodegenerative diseases occur when nerve cells in the brain or peripheral nervous system progressively lose structure and/or function and ultimately die. While significant challenges remain in research and drug development for rare neurodegenerative diseases, meaningful progress has been made in several of these diseases. A comprehensive characterization of the molecular mechanisms that cause the underlying pathophysiology will result in our ability to identify targets for effective therapeutic options, but such advances need to be translated into actionable solutions that can accelerate and optimize the design of clinical trials to evaluate novel therapies. This includes quantification of disease pathogenesis and natural history, quantifying disease progression, evaluation of actionable biomarkers, and the generation of regulatory-grade tools that can accelerate research and drug development.
“The effects of rare neurodegenerative diseases are devastating, with very few effective therapeutic options available to patients,” stated U.S. Food and Drug Commissioner Robert M. Califf, M.D. “We recognize the urgent need for new treatments that can both improve and extend the lives of people diagnosed with these diseases.”
To transform these challenges into opportunities, collaboration, fueled by data, will be key.
A New Public-Private Partnership
Announced by FDA and NIH, and convened by C-Path, the new public-private partnership (PPP), Critical Path for Rare Neurodegenerative Disease (CP-RND), will bring together multiple experts in rare neurodegenerative diseases, including ALS, as well as private entities, patient communities and advocacy organizations to accelerate and advance our understanding of disease pathology, treatment options, diagnostics, and drug development. C-Path has built a robust infrastructure within a precompetitive framework that provides a safe harbor for commercial drug developers to bring data and shared learnings. This, combined with independent efforts from the FDA and NIH, provides a prime opportunity and unique global venue for accelerating medical product development in rare neurodegenerative diseases. CP-RND will also facilitate interactions and collaboration with the FDA and NIH and the external biomedical research community for the consideration of cross-cutting clinical and regulatory policy issues, and consistent decision making.
Three main purposes for this effort, and its impact on rare neurodegenerative diseases, including ALS, are to:
- Establish a transformative PPP that catalyzes on all aspects of scientific discovery and drug development
- Develop an individual and experiment-level integrated database
- Generate actionable solutions that can tangibly accelerate drug development
The FDA announced the PPP in a press release on Sept. 14, 2022.
How CP-RND Will Work
CP-RND will help increase our understanding of disease pathogenesis and natural history by quantifying disease progression including evaluation of potential biomarkers from patient-level data, optimization of clinical trial design including exploration of innovative trial design and improving the efficiency of drug development.
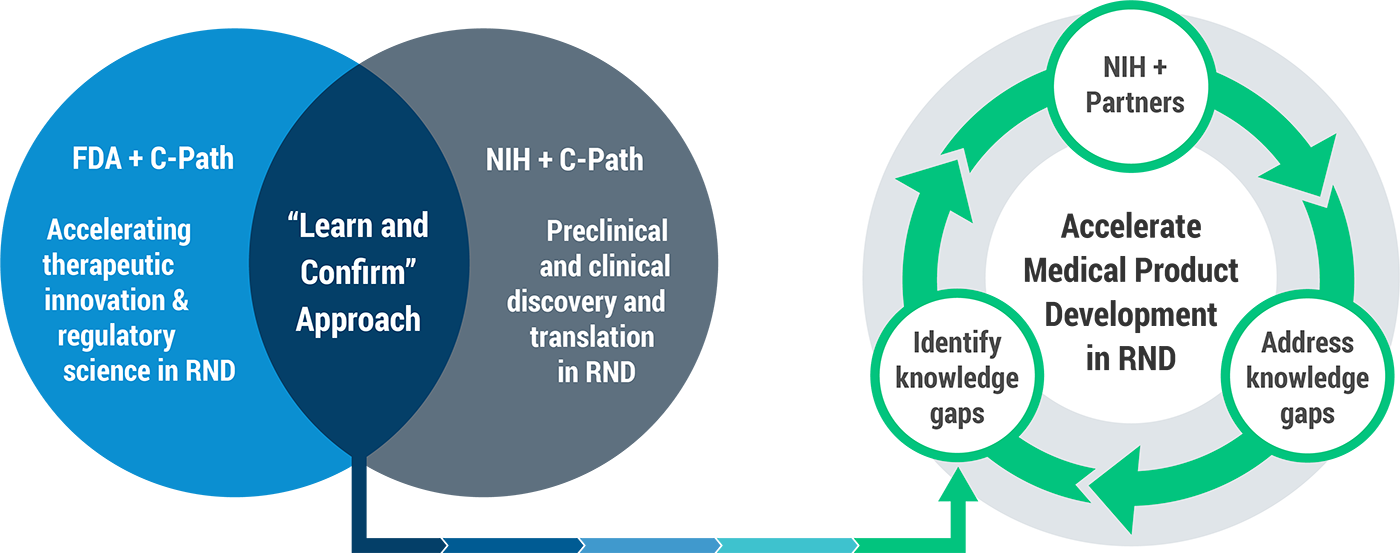
The partnership will benefit from the extensive experience that C-Path has accrued in its more than 17 years in operation. The institute has coordinated unique PPPs across more than 20 therapeutic areas; including comprehensive collaborations that have helped transform the drug development landscape in areas beyond neuroscience, such as polycystic kidney disease and tuberculosis.
C-Path’s proven proficiencies will be leveraged to ensure the success of this effort, specifically the organization’s track record in generating tangible solutions that have accelerated drug development in numerous diseases. This, together with the capabilities of its Rare Disease Cures Accelerator-Data and Analytics Platform (RDCA-DAP ®) to integrate multiple patient-level data sources across rare diseases, will provide the foundation for leveraging advances in basic and clinical sciences, supported by NIH, and innovative regulatory science, supported by FDA.
C-Path will leverage its knowledge and experience in identification and generation of consensus on key unmet needs by incorporating perspectives from relevant stakeholders across the patient community, academic community, regulatory agency, and industry, to aggregate patient-level data (clinical trial, natural history, and relevant real-world data) for the generation of drug development tools and solutions, identification and evaluation of promising biomarker candidates (fluid, imaging, digital), consideration of reasonably likely surrogate endpoint candidates, and evaluation of outcome assessments (patient-reported outcome (PRO) measures, electronic clinical outcome assessment (eCOA) measures, functional assessment scales, etc.)
Interested in participating in this collaboration? Please inquire and include your contact information at
CP-RND@c-path.org.

